JEPonline
Journal
of
Exercise
Physiologyonline
ISSN
1097-9751
An
International Electronic
Journal
for Exercise Physiologists
Vol 2 No 1 January 1999
|
Nutrition
and Exercise
Glycerol
hyperhydration alters cardiovascular and renal function
P. MONTNER1,2,
Y. ZOU3, R.A. ROBERGS
4, G. MURATA1,2, D. STARK1,
C. QUINN1, S. WOOD5,
D. LIUM1, and E.R. GREENE3
1Department
of Medicine, University of New Mexico School of Medicine, Albuquerque,
NM, 87131 , 2The Albuquerque Veterans Affairs Medical
Center, Albuquerque, NM, 87108,
3School of Science and
Engineering, New Mexico Highlands University, Las Vegas, NM 87143, 4The
Center for Exercise and Applied Human Physiology, University of New Mexico,
Albuquerque, NM, 87131,
5 Highlands University, Las Vegas, NM,
87701
P. MONTNER, Y. ZOU, R.A.
ROBERGS, G. MURATA, D. STARK, C. QUINN, S. WOOD, D. LIUM, and
E.R. GREENE. Glycerol hyperhydration alters cardiovascular
and renal function.JEPonline
Vol
2 No 1 1999.
Six subjects randomly
completed four experimental trials consisting of 2 hours of controlled
fluid ingestion at rest, followed by 110 min of cycle ergometry exercise
at 44 ± 11 %VO2max. Fluid was also
ingested during exercise, and the trials differed in either the pre-exercise
or during exercise fluid ingestion. The control trial consisted of the
pre-exercise ingestion of 26 mL/kg of flavored water, and 5 mL/kg every
20 min of 5% glucose during exercise (WC). For the remaining trials the
solutions ingested were 1.2 g/kg glycerol in a total of 26 mL/kg (GH) pre-exercise,
and either 5% glucose (GC), 0.5% glycerol in 5% glucose (GCGA), or 1.5%
glycerol in 5% glucose (GCGB) during exercise. Compared to WC, GH decreased
urine flow (4.7 ± 2.2 vs 7.6 ± 3.7 mL.min, p<0.001) and
free water clearance (-1.4 ± 1.3 vs 2.5 ± 1.3 mL/min, p<0.001).
Consequently, pre-exercise hydration was largest with GH (0.9 ±
0.4 vs 0.3 ± 0.3 L for the mean of GC, GCGA and GCGB vs WC). Compared
to WC, GH also increased serum osmolality (283.3 ± 3.3 to 281.0
± 2.6 vs 284.3 ± 0.2 to 291.0 ± 2.3 mOsmol/kg, p<0.01)
despite no difference in serum antidiuretic hormone (ADH) (2.3 ±
2.0 vs 2.4 ± 1.0 pg/mL at 120 min). When comparing WC to GCGA and
GCGB, continued glycerol ingestion during exercise increased cardiac stroke
volume (163.3 ± 27.9 vs 174.4 ± 22.8 mL, p<0.01), and
decreased heart rate (128.2 ± 19.0 vs 122.0 ± 14.5 b/min,
p<0.01). GH increases body hydration by decreasing renal free water
clearance via a non-ADH mediated mechanism. Continued glycerol and water
ingestion during exercise increases the cardiovascular benefits of pre-exercise
GH, and prolongs the state of hyperhydration.
Key Words: OSMOLALITY,
ANTIDIURETIC HORMONE, FREE WATER CLEARANCE, STROKE VOLUME
Introduction
Compared to the ingestion of an equal
volume of water, ingestion of glycerol and water providing approximately
1.0 g/kg body weight of glycerol in a total volume of 20 to 26 mL/kg body
weight (glycerol hyperhydration, GH) can significantly decrease urine volume
and cause a fluid retention of between 300 to 730 mL (1-4).
GH has also been shown to improve evaporative cooling during exercise in
a hot environment (2), and prolong time to fatigue during
endurance exercise (3). The combination of the pre-exercise
ingestion of glycerol, followed by added glycerol ingestion during exercise
has not been studied. However, Koenigsberg et al. (5)
showed that compared to the ingestion of water, continued glycerol and
water ingestion can maintain an increased body hydration for up to 40 hours.
The increase in body water from GH is known to occur without an increase
in plasma ADH (4), and has been theorized to result from
a glycerol mediated increase in water reabsorption in the distal tubules
and collecting ducts of the kidney (4,6).
Since previous studies (7-10)
of fluid infusion to expand the plasma volume found increases in stroke
volume and lowered heart rates, GH may also provide benefits to cardiovascular
function during exercise. For example, glycerol ingestion has been shown
to lower heart rates during exercise in the heat (2),
yet contradictory evidence exists for an expansion of plasma volume following
GH (1,2,4,11).
Prior research has mainly compared pre-exercise
glycerol hyperhydration to an equal volume of saline (1),
orange juice (2) or water ingestion (3,4).
The generalizability of using water as a control solution can be questioned
due to the known superiority of carbohydrate-electrolyte solutions over
water for sustaining hydration during exercise (12),
and for improving rehydration from dehydration (13).
Based on our summary of prior research,
it was our intent to compare pre-exercise GH to water ingestion. As most
athletes ingest carbohydrate beverages during exercise, we also wanted
to compare pre-exercise GH to GH followed by liquid carbohydrate ingestion
during exercise. We hypothesized that the hyperhydration induced by GH
would be larger than from water, and better sustained during exercise with
additional glycerol ingestion. We also hypothesized the GH and continued
glycerol ingestion would retain better hydration and cardiovascular benefits
compared to GH and liquid carbohydrate ingestion during exercise. Based
on the findings of Freund et al. (4), we further hypothesized
that hyperhydration from GH would occur without increases in serum ADH,
yet still be associated with a reduction in free water clearance.
Materials and Methods
Subjects and Approval
Six subjects, four men and two women,
volunteered to participate. The female subjects were eumenorrheic and were
studied in the follicular phase of their menstrual cycle, as suggested
by verbal interview and verified by a serum progesterone concentration
<5 ng/mL. The study was approved by the Human Research Review Committees
of each agency involved in this research.
Procedures
Subjects were first familiarized with
the exercise equipment, and a preliminary cycle ergometry test to VO2max
was completed to determine relative submaximal workloads for the subsequent
exercise trials. For all testing, exercise was performed on a semi-recumbent
cycle ergometer, ambient conditions were maintained at 23.5 - 24.5 °C
and 25 - 27% humidity, and the altitude was 1600 m.
Subjects completed four trials in a randomized,
double blind procedure. Trials were performed at least one week apart,
and at the same time of day. Subjects were admitted to a National Institutes
of Health supported Clinical Research Center 48 hours prior to each trial.
During this period, identical self-selected diets were given to each individual
at each admission, and training activities were curtailed. After breakfast
on the morning of each trial, subjects were transferred to the exercise
testing facility. After arrival, a nude, post-void weight (± 0.1
kg) (Pennsylvania 3000 Electronic Scale) for each subject was recorded
and baseline urine specimens were obtained. An antecubital vein heparin
lock (18 gauge) was placed, and subjects remained in a seated position
for 10 min, after which venous blood specimens (7 mL) were obtained.
For the control hydration condition (WC),
26 mL/kg body weight of aspartame sweetened and artificially flavored water
was ingested over a 2 hour period before exercise and followed by 5% CHO
(5 mL/kg body weight every 20 min) during exercise (Table
1). For the GH trials, subjects ingested 1.2 gm/kg body weight
of glycerol in 26 mL/kg of solution over a 2 hour period before exercise,
and every 20 min during exercise ingested 5 mL/kg body weight of 5% CHO
(GC), 5% CHO in 0.5% glycerol (GCGA), or 5% CHO in 1.5% glycerol (GCGB).
The glucose solutions ingested during exercise also had 20 mEq/L of sodium
chloride, and all solutions were kept at 4°C. Pre-exercise GH involved
the ingestion of a bolus volume of concentrated glycerol (200 g/L), providing
1.0 g/kg body weight during the initial 30 min, as described in detail
by Montner et al. (3). Thereafter, water was ingested
every 30 min to 120 min, and at 60min the remaining glycerol (0.2 g/kg)
was ingested. The total volume of water ingestion was the balance of 26
mL/kg body weight minus the volume of the glycerol solution.
Table 1. Constituents
of the different drinks
| Drink |
Glycerol
(g/kg) |
Glucose
(g/100mL) |
Electrolytes
(mEq/L-NaCI) |
| Glycerol Bolus |
1.2 |
0 |
0 |
Glucose Solutions
During Exercise |
|
|
|
| GC |
0 |
5.0 |
20.0 |
| GCGA |
0.5 |
5.0 |
20.0 |
| GCGB |
1.5 |
5.0 |
20.0 |
During the 2 hour hydration period, subjects
maintained a sitting position. All urine during this period was collected,
the volume measured, and samples from baseline and 120 min were used for
analyses of creatinine and osmolality. Following the 2-hour pre-exercise
hydration period, the subjects performed 110 min of exercise in a semi-recumbent
position on a cycle ergometer (Model 846T, Quinton Instrument Co., Seattle
WA) at 44 ± 11% V02max. This workload
and position was selected to ensure that subjects could complete the exercise
duration, and allow for more easily determined and accurate stroke volume
measurements using Doppler flowmetry. Venous specimens for blood elastic
yield stress (EYS) were taken every 40 min, and at 60 and 120 min of rest
for serum osmolality and ADH. Serum glycerol was determined at baseline,
at 120 min of rest and after 110 min of exercise. Nude body weight was
also measured at the end of hydration (120 min), and after 110 min of exercise
each subject towel dried and was again weighed nude.
Analytical Procedures and Calculations
Indirect Calorimetry
The exercise protocol used to determine
V02max consisted of 1 min duration increments
at between 20 to 30 Watts/min to ensure a total test duration between 10
to 15 min. Expired gas analysis was completed using a time averaged automated
system (Incarepulmobil, Erich Jaeger, Rockford, IL) involving volume measurement
with a heated pneumotach. Expired fractions of oxygen and carbon dioxide
were determined using oxygen (zirconian cell) and carbon dioxide (infra-red)
analyzers. Prior to each test the pneumotach was calibrated with seven
volumes of a 1 LATPS calibrated syringe, and analyzers were calibrated
to room air and medically certified gasses (100% nitrogen; and 15% O2,
5% CO2, balance nitrogen).
Central Cardiovascular Hemodynamics
Stroke volume was determined by pulsed
Doppler (PD) flowmetry (Medsonics, Fremont, CA) utilizing a suprasternal
approach (14-17). This standard noninvasive method has
been described in detail and validated by us in previous studies (14,15)
and by others (16,17). In our hands,
the 95% confidence levels for precision (percent changes) and accuracy
(absolute values) are ± 7% and ± 13% respectively. The measurements
were taken with a 2.0 MHz transducer at time 0, 45, and 120 min of the
pre-exercise period, and at the following intervals of the exercise: 10,
30, 50, 70, 90 and 110 min. Heart rate was determined from PD waveform
intervals. The PD data were subsequently processed by blinded observers
to yield data of Doppler angle, aortic diameter and mean blood flow velocity.
Pulse volume was calculated using the Doppler equation.
Blood and Urine Assays
Blood and urine samples were assayed in
duplicate for creatinine (calorimetric method, IL Monarch 2000, Lexington,
MA, or reflectometric method, Kodak Ecktachem 700XR, Rochester, NY), and
osmolality (freeze point depression, Nichols Inst., CA). Serum ADH was
assayed by radioimmunoassay (Nichols Inst., CA) with a test-retest coefficient
of variation <5%. Glycerol was assayed by enzymatic spectrophotometry
(Boehringer Diagnostics, CA), and EYS was quantified as a marker of blood
viscosity (Vilastic 3, Vilastic Scientific, Inc).
Renal Function
Renal free water clearance was calculated
as urine flow - ((urine osmolality x flow) / serum osmolality). Creatinine
clearance was calculated as ((urine creatinine x flow) / serum creatinine).
Statistics
Due to concerns over statistical power,
descriptive statistics were first completed on all variables to determine
mean differences, variance and effect sizes. For the variable of stroke
volume, determined to be the most difficult to detect significance for
during exercise with 6 subjects per group, the mean difference between
WC and GCGB at 50 min was 15.2 mL (162.8 ± 26.5 vs 178.0 ±
16.2 mL), with an effect size of 0.71 and a b error = 0.85 from a one-tailed
t-test (Statmate, Graphpad Software, San Diego CA). To improve statistical
power, we decided to conduct statistical analyses from specific planned
comparisons after collapsing specific trials to compare water vs GH, and
glycerol ingestion during exercise to CHO ingestion (Statistica, Statsoft
Inc., Tulsa OK). Due to repeat analyses of means and apriori protection
against type II errors, a Bonferoni adjustment was used to protect against
type I errors, resulting in statistical acceptance at p = 0.017 (0.05/3).
This approach enabled us to improve statistical power, as indicated by
the comparison of stroke volume data of WC (N = 36) compared to the combined
data from GCGA and GCGB (N = 60) throughout exercise (mean difference =
11.4 mL, effect size = 0.4, b error = 0.3). Data are presented as mean
± SD.
Results
Subjects
Subjects trained regularly with cycling
or running and had a mean age, body mass, and maximal oxygen uptake (VO2max)
of 27 ± 3 yr., 68 ± 6 kg, and 55.0 ± 2.8 mL·kg-1
min-1 respectively.
Pre-exercise Hydration
Due to the need to perform apriori statistical
analyses, group mean data for pre-exercise GH represent the combined data
from GC, GCGA and GCGB trials. Pre-exercise water ingestion did not change
serum osmolality (283.3 ± 3.3 to 281.0 ± 2.6 mOsmol/kg).
However, pre-exercise GH significantly increased serum osmolality at 120
min compared to baseline (284.3 ± 0.2 to 291.0 ± 2.3 mOsmol/kg,
for WC vs GH trials, p<0.01), but did not increase serum ADH (2.3 ±
2.0 vs 2.4 ± 1.0 pg/mL at 120 min for WC vs GH trials). Pre-exercise
GH significantly increased serum glycerol at 120 min compared to baseline,
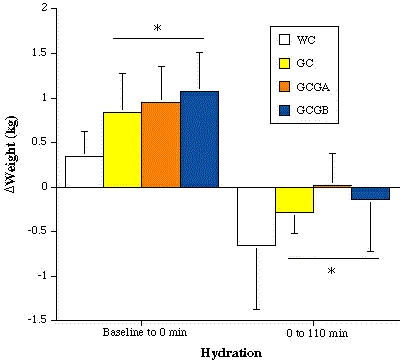
and compared to WC (Figure 1). Body weight
gain at 120 min was significantly larger with GH (Figure
2), resulting in an additional 600 mL of water storage (p = 0.0015).
These body weight changes coincided with significant reductions in urine
flow and free water clearance for WC compared to GH (Figure
3), but no change in creatinine clearance (133 ± 35 vs 110
± 45 mL/min for WC and GH trials). EYS results did not differ between
trials, and the mean values during pre-exercise hydration were 0.12 ±
0.02 dyne/cm2 for both WC and GH.
Figure
1. The change in serum glycerol over time and during each trial.
* = p<0.01 from WC and
baseline, # = p<0.01 from GC and GCGA.

Exercise Hydration
During the GH trials, blood glycerol remained
elevated above WC during exercise. Continued ingestion of glycerol in the
GCGA and GCGB trials did not cause a detectable increase in serum glycerol
until after 110 min of exercise, when serum glycerol in the GCGB trial
was significantly larger compared to all other trials (Figure
1).
Pre-exercise GH and the continued ingestion
of glycerol and liquid carbohydrate during exercise (GC, GCGA, GCGB) significantly
(p<0.01) improved body weight maintenance (retained hydration at 110
min of exercise compared to baseline) compared to WC (Figure
2). Mean SV during exercise was significantly larger for the trials
involving added glycerol ingestion (GCGA and GCGB) compared to WC and GC,
and mean HR was significantly lower in GCGA and GCGB than WC and GC (Figures
3 & 4).
Figure
2. The change in body weight from baseline to 120 min pre-exercise
(percentage change in weight), and from baseline to 110 of exercise during
each trial. * = p<0.01 for WC vs GC, GCGA and GCGB.
Discussion
As in the previous studies of Riedesel
et al. (1), Lyons et al. (2), Montner
et al. (3) and Freund et al. (4), we
found that GH, compared with water ingestion, resulted in an increased
fluid retention. Furthermore, as with the study of Freund et al. (4)
we documented that GH increased osmolality, reduced free water clearance,
but did not increase plasma ADH. Thus, glycerol appears to be an ineffective
osmole for ADH, like urea and glucose (16). Murray et
al. (11) found that ADH increased more from baseline
with glycerol ingestion during exercise (3.2 to 9 pg/mL) compared to water
ingestion alone (3.2 to 6 pg/mL). However, Murray et al. (11)
did not follow a pre-exercise GH procedure, thereby preventing a glycerol
hyperhydration which others have shown requires an approximate 2 hour pre-exercise
hydration regimen (1-4). It remains unclear why the protocol
of Murray et al. (11) caused an increase in blood ADH.
We also reported that pre-exercise GH and
glycerol ingestion during exercise better preserved body hydration during
exercise compared to water and liquid carbohydrate ingestion (Figure
1). This finding supports the research of Lyons et al. (2),
who documented that GH provided more fluid for sweat output during exercise
in dry heat compared to water/orange juice, without further compromising
body water stores. It appears that GH enables the body to tolerate water
loss better by simply providing more fluid rather than by decreasing fluid
loss during exercise/heat stress.
The causal mechanism of the body water
gain from GH has not been clearly established. However, the mechanism for
the hyperhydration afforded by GH has been theorized to result from the
increased glycerol reabsorption in the kidney which also induces an increased
water reabsorption (4,6,18).
Due to the high diffusive properties of glycerol, the sustained elevation
in blood glycerol causes glycerol and water to be evenly distributed throughout
the body water spaces external to the brain and eyes (18).
It is therefore logical to assume that the hydration benefits of GH are
an expansion of the intra-cellular, interstitial, and vascular body water
compartments (18). Riedesel et al. (1)
hypothesized that as glycerol was metabolized or excreted, the increased
volume of body water would become available for metabolic use. This remains
a viable explanation for the eventual benefits of GH to cardiovascular
and thermoregulatory capacities during exercise.
Although we did not quantify plasma volume
in this study, an improved retention of plasma volume during exercise (less
hemoconcentration) is a possible explanation for our documented increases
in cardiac stroke volume. Based on this interpretation, we feel it is important
to identify some difficulties in detecting a plasma volume expansion following
GH. Despite the theoretical rationale for GH to increase all body water
compartments (18), there are contradictory findings
of a plasma volume expansion following glycerol ingestion. Gleeson et al.
(19) found that 400 mL of a 1 g/kg glycerol solution
increased PV by 10% during pre-exercise hydration while placebo and glucose
feeding did not. PV changes were calculated from changes in hemoglobin
and hematocrit as described by Dill and Costill (20).
With ingestion of similar amounts of glycerol and water during exercise,
Murray et al. (11) also found that glycerol maintained
PV better than a water placebo or glucose solution. Conversely, Freund
et al. (4) reported no differences in blood and plasma
volumes following glycerol vs water ingestion, despite the 500 mL increase
in total body water with glycerol ingestion.
The difficulty in detecting an increase
in plasma volume after GH is not surprising, as the largest increase in
fluid retention seen with GH has been 730 mL over a 2 hour period (3).
This time frame would allow the glycerol and water to more evenly diffuse
throughout the total body water As plasma volume is the smallest of the
body water compartments, a 730 mL increase in total body water evenly distributed
among body water compartments would cause an approximate 45 mL increase
in plasma volume, which would be difficult to detect given the errors of
dye dilution, and the relatively small numbers of subjects used in past
research (1-4).
Interestingly, the significant findings
for an expansion of plasma volume by Gleeson et al. (19)
and Murray et al. (11) may be related to the decreased
time provided to subjects after glycerol ingestion (45 min and 0 min, respectively).
This shorter time frame may have prevented an equilibration of the glycerol
and water load throughout the total body water volume, resulting in a greater
plasma volume expansion.
No previous published studies have directly
measured SV after GH. In the present study, SV was increased with continued
ingestion of glycerol during exercise compared to liquid CHO ingestion
after water or pre-exercise GH (Figure 3).
Figure
3. The decreases in pre-exercise urine flow and free water clearance,
averaged over 2 hours, for the WC and GH (GC, GCGA, GCGB) trials. * = p<0.01
for WC vs GH.

These results coincided with reciprocal
changes in heart rate (Figure 4). These
functional measurements may be physiologically more significant than the
difficulty in documenting a change in plasma volume after GH, as previously
explained. The cardiovascular benefit of pre-exercise GH may not be in
the absolute increase in plasma volume, but the potential for using added
extravascular water stores to better maintain plasma volume during exercise/heat
stress.
Figure
4. The changes in heart rate (HR) and stroke volume (SV) during
pre-exercise and exercise hydration periods of each trial. For HR,
* = p<0.01 for all trials from baseline (-120 min). # = p<0.01 for
all of exercise for WC and GC vs GCGA and GCGA and GCGB. For SV,
* = p<0.01 for all trials from baseline (-120 min). # = p<0.01 for
all of exercise for WC and GC vs GCGA and GCGB.

In a 1982 study of plasma volume expansion
by Kanstrup and Ekblom (10), HR was an average of 5
bpm lower than control at submaximal exercise intensities. Furthermore,
Fortney et al. (7) infused 533 mL of isotonic albumin
and lactated Ringers solution before exercise, and found that HR during
exercise was 3-6 bpm lower than control and SV was an average of 13 mL
higher than control. Hopper et al. (9) found that during
submaximal cycle ergometry, infusion of 403 mL of dextran solution increased
SV by 11% (14 ml), CO by 7% (1.32 L/min), and decreased HR from 141 bpm
to 138 bpm. The HR and SV findings of our study are in the range found
in these studies involving artificial PV expansion.
Another important finding of this study
was the added benefit of glycerol ingestion during exercise to pre-exercise
GH. Surprisingly, pre-exercise GH did not reveal results during exercise
that were significantly different from water ingestion followed by liquid
carbohydrate (Figures 2 and
3). These findings differ to the results of Montner et al. (3)
and Lyons et al. (2). However, the exercise intensity
of Montner et al. (3) was larger (74% VO2max
to exhaustion) than the present study, and the subjects of Lyons et al.
(2) were required to exercise in a hot environment (90
min at 60% VO2max in 42°C). Perhaps
the maximal potential for benefits from pre-exercise GH are observed during
more severe exercise and/or heat stress.
The benefit of continued glycerol and water
ingestion during exercise is supported from data of glycerol removal after
pre-exercise GH. Robergs and Griffin (18) have calculated
that the rate of glycerol removal from the body after GH approximates 30-50
g/hr. Thus, two hours after GH more than 50% of the glycerol ingested has
been removed from the body via urinary excretion and metabolism. Based
upon the direct action of glycerol on water reabsorption in the distal
tubule, the continued retention of added body water would require a sustained
elevation in blood glycerol. This result was clearly seen in the blood
glycerol data of the GCGB trial after 110 min of exercise (Figure
1).
Conclusions
Pre-exercise GH resulted in fluid retention
by reducing renal free water clearance, but through a non-ADH mediated
mechanism. Compared to pre-exercise water or GH followed by liquid carbohydrate
ingestion, pre-exercise GH followed by continued glycerol and water ingestion
during exercise increases stroke volume and lowers heart rate during exercise.
These additional cardiovascular benefits of GH during exercise occurred
with an additional 5 g glycerol/hr in 350 mL/hr. It remains unclear whether
larger amounts of glycerol ingestion during exercise would further sustain
improved hydration, and whether these responses during recumbent low to
moderate intensity cycling could relate to improved exercise performance
at higher relative exercise intensities.
ACKNOWLEDGMENTS:
This
study was supported in part by the National Institutes of Health General
Clinical Research Center award no. 5M01RR00997-16, the Dedicated Health
Research Funds of the University of New Mexico School of Medicine, the
Albuquerque Veterans Affairs Medical Center, and the Center For Exercise
and Applied Human Physiology - the University of New Mexico. We would like
to thank the staff of the Albuquerque VAMC Medical Media Service and Mary
Hill of the AVAMC for their help in preparing this manuscript.
Address correspondence to:
Paul Montner, M.D., M.S. AVAMC 2100 Ridgecrest Dr. SE Albuquerque, NM 87108
(505) 265-1711 x 4540 FAX (505) 256-5751 Glycerol Hyperhydration Alters
Cardiovascular and Renal Function
References
1. Riedesel
ML, Allen DY, Peake GT, Al-Qattan K. Hyperhydration with glycerol solutions.
J
Appl Physiol. 1987;63:2262-2268.
2. Lyons
TP, Riedesel M, Meuli LE, Chick TL. Effects of glycerol induced hyperhydration
prior to exercise in the heat on sweating and core temperature. Med
Sci Sports Exerc. 1990;22:477-483.
3. Montner
PK, Stark DM, Riedesel ML, Murata GA, Robergs RA, Timms M, Chick TL. Pre-exercise
glycerol hydration prolongs cycling endurance time. Int J Sports Med.
1996;17:27-33.
4. Freund
BJ, Montain SJ, Young AJ, Sawka MN, DeLuca JP, Pandolf KB, Paleri CR Glycerol
hyperhydration: hormonal, renal, and vascular fluid responses. J Appl
Physiol. 1995;79:2069-2077.
5. Koenigsberg
PS, Martin KK, Hlava HR. Sustained hyperhydration with glycerol ingestion.
Life
Sci. 1995;5:645-653.
6. Kruhoffer
P, Nissan OI. Handling of glycerol in the kidney. Acta Physiol Scand.
1963;59:284-294.
7. Fortney
SM, Wenger CB, Bove JR, Nadel ER. Effect of blood volume on forearm venous
and cardiac stroke volume during exercise. J Appl Physiol. 1983;55:884-890.
8. Krip
B., Gledhill N, Jamnik V, Warburton D. Effect of alterations in blood volume
on cardiac function during maximal exercise. Med. Sc. Sports Exerc.
1997;29:1469-1476.
9. Hopper
MK, Coggan AR, Coyle EF. Exercise stroke volume relative to plasma-volume
expansion. J Appl Physiol. 1988;64:404-408.
10. Kanstrup
IL, Ekblom B. Acute hypervolemia, cardiac performance, and aerobic power
during exercise. J Appl Physiol. 1982;52:1186-1191.
11. Murray
R, Eddy DE, Paul GL, Seifert JG, Halaby GA. Physiological responses to
glycerol ingestion during exercise.
J Appl Physiol. 1991;71:144-149.
12. Below
PR, More-Rodriguez R, Gonzales-Alonso J, Coyle EF. Fluid and carbohydrate
ingestion independently improve performance during 1 h of intense exercise.
Med.
Sci. Sports Exerc. 1995;27:200-210.
13. Gisolfi
CV, Duchman SM. Guidelines for optimal replacement beverages for different
athletic events. Med Sci Sports Exerc. 1992;24:679-687.
14. Loeppky
JA, Greene ER, Hoekenga DE, Caprihan A, Luft UC. Beat-by beat stroke volume
assessment by pulsed Doppler in upright and supine exercise. J Appl
Physiol. 1981;50:1173-1182.
15. Shaw
JC, Johnson EC, Boyles WF, Greene ER. Noninvasive Doppler determination
of cardiac output during submaximal and peak exercise. J Appl Physiol.
1985;59:722-731.
16. Innes
JA, Simon TD, Murphy K, Guz A. The effects of exercise and subject age
on pulsed Doppler measurements of left ventricular ejection in normal man.
Q
J Exp Physiol. 1988;73:323-341.17. Spencer KT, Laing
RM, Newman A, Barow KM, Shroff SG. Doppler and electromagnetic comparisons
of instantaneous aortic flow characteristics in primates. Circ Res.
1991;68:1369-1977.
18. Robergs
RA, Griffin SE. Glycerol ingestion: Clinical and applied applications.
Sports
Medicine. 1998;26:145-167.
19. Gleeson
M, Maughan RJ, Greenhaff PL. Comparison of the effects of pre-exercise
feeding of glucose, glycerol and placebo on endurance and fuel homeostasis
in man. Eur J Appl Physiol. 1986;55:645-653.
20. Dill
DB, Costill DL. Calculation of percentage changes in volumes of blood,
plasma, and red cells in dehydration.
J Appl Physiol. 1974;37:247-248.
Copyright ©1999
American
Society of Exercise Physiologists
All Rights Reserved
ASEP
Table of Contents
January
JEPonline (home page)